1.The existence of a substance in more than one solid
modifications is known as
a) Isomorphism
b) Polymorphism
c) Amorphism
d) Allotropy
Explanation: Polymorphisms
2. The second order Bragg diffraction of X-rays with \[\lambda =1.0Å\] from a set of parallel planes in a metal occurs at an angle of
60°. The distance between the scattering planes in the crystal
is
a) 0.575Å
b) 1.00Å
c) 2.00Å
d) 1.15Å
Explanation:
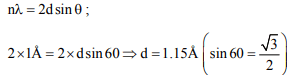
3. Superconductors are derived from compounds of
a) p-block elements
b) lanthanides
c) actinides
d) transition elements
Explanation: Superconductors are derived from compound of p block elements.
4.If NaCl is doped with \[10^{-4}\] mol % of \[SrCl_{2}\] , the concentration
of cation vacancies will be \[\left(N_{a}=6.02 × 10^{23}mol ^{-1}\right)\]
a) \[6.02 × 10^{16}mol ^{-1}\]
b) \[6.02 × 10^{17}mol ^{-1}\]
c) \[6.02 × 10^{14}mol ^{-1}\]
d) \[6.02 × 10^{15}mol ^{-1}\]
Explanation:

5. The fraction of total volume occupied by the atoms present
in a simple cube is
a) \[\frac{\pi}{3\sqrt{2}}\]
b) \[\frac{\pi}{4\sqrt{2}}\]
c) \[\frac{\pi}{4}\]
d) \[\frac{\pi}{6}\]
Explanation:
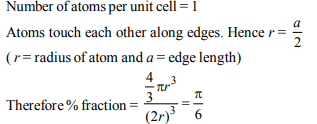
6.If ‘a’ stands for the edge length of the cubic systems : simple
cubic, body centred cubic and face centred cubic, then the
ratio of radii of the spheres in these systems will be
respectively,
a) \[\frac{1}{2}a :\frac{\sqrt{3}}{4}a:\frac{1}{2\sqrt{2}}a\]
b) \[\frac{1}{2}a :\sqrt{3}a:\frac{1}{\sqrt{2}}a\]
c) \[\frac{1}{2}a :\frac{\sqrt{3}}{2}a:\frac{\sqrt{3}}{2}a\]
d) \[1a :\sqrt{3}a:\sqrt{2}a\]
Explanation:

7. With which one of the following elements silicon should be
doped so as to give p-type of semiconductor ?
a) Germanium
b) Arsenic
c) Selenium
d) Boron
Explanation: The semiconductors formed by the introduction of impurity atoms containing one elecron less than the parent atoms of insulators are termed as p-type semiconductors. Therefore silicon containing 14 electrons is to be doped with boron containing 13 electrons to give a p-type semi-conductor.
8. AB; crystallizes in a body centred cubic lattice with edge
length ‘a’ equal to 387 pm. The distance between two
oppositely charged ions in the lattice is :
a) 335 pm
b) 250 pm
c) 200 pm
d) 300 pm
Explanation:
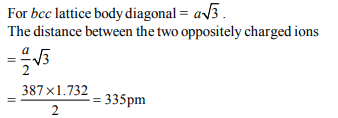
9. Among the following which one has the highest cation to
anion size ratio?
a) NaF
b) CsI
c) CsF
d) LiF
Explanation:

10. A solid compound XY has NaCl structure. If the radius of the
cation is 100 pm, the radius of the anion \[\left(Y^{-}\right)\] will be :
a) 275.1 pm
b) 322.5 pm
c) 241.5 pm
d) 165.7 pm
Explanation:
