1. Crystals can be classified into basic crystal habits, equal to
a) 7
b) 4
c) 14
d) 2
Explanation: Seven crystal systems
2. Tetragonal crystal system has the following unit cell
dimensions
a) \[a = b = c, \alpha = \beta = \gamma = 90°\]
b) \[a = b \neq c, \alpha = \beta = \gamma = 90°\]
c) \[a\neq b \neq c, \alpha = \beta = \gamma = 90°\]
d) \[a = b \neq c, \alpha = \beta = 90° \gamma = 120°\]
Explanation:
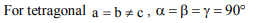
3. A match box exhibits
a) cubic geometry
b) monoclinic geometry
c) tetragonal geometry
d) orthorhombic geometry
Explanation:
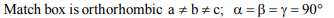
4. When molten zinc is cooled to solid state, it assumes hcp
structure. Then the number of nearest neighbours of zinc
atom will be
a) 4
b) 6
c) 8
d) 12
Explanation: Number of nearest neighbours means coordination number = 12
5. Co-ordination number of \[Na^{+}\] in NaCl is
a) 4
b) 3
c) 6
d) 5
Explanation: Coordination number 6
6. A crystal lattice with alternate +ve and –ve ions has radius
ratio 0.524. Its co-ordination number is
a) 4
b) 3
c) 6
d) 12
Explanation:

7. How many unit cells are present in a cube-shaped ideal
crystal of NaCl of mass 1.00 g?
[Atomic masses : Na = 23, Cl = 35.5]
a) \[2.57 × 10^{21}\] unit cells
b) \[5.14 × 10^{21}\] unit cells
c) \[1.28 × 10^{21}\] unit cells
d) \[1.71 × 10^{21}\] unit cells
Explanation:
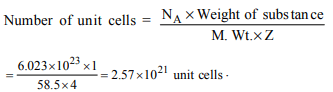
8. Which of the following is example of body centred
cube ?
a) Mg
b) Zinc
c) Copper
d) Sodium
Explanation: Na has Body centred cubic lattice.
9. Which of the following describes the hexagonal close packed
arrangement of spheres?
a) ABCABA
b) ABCABC
c) ABABA
d) ABBABB
Explanation: ABAB is hexagonal close packing
10. A metallic crystal crystallizes into a lattice containing a
sequence of layers AB AB AB......Any packing of spheres
leaves out voids in the lattice. What percentage of volume
of this lattice is empty space?
a) 74%
b) 26%
c) 50%
d) none of these
Explanation: In AB AB packing spheres occupy 74%. 26% is empty.