1. Which of the following is a man made element?
a) Ra
b) Fr
c) Rn
d) Lr
Explanation:
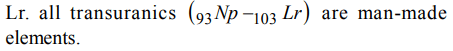
2. Which one of these is most reactive ?
a) Na
b) K
c) Pb
d) Mg
Explanation: K is most reactive
3. Alkali metals are generally extracted by
a) reduction methods
b) double decomposition methods
c) displacement methods
d) electrolytic methods
Explanation: Alkali metals are highly electropositive and thus highly reducing. Therefore reduction , double decomposition and displacement methods for their extraction are not suitable. Only electrolytic methods are useful for their extraction.
4. The alkali metal which is a liquid at room temperature is
a) Fr
b) Cs
c) Rb
d) Na
Explanation: Fr
5. The alkali metal which can emit its outermost electron under the influence of even candle light is
a) Na
b) Rb
c) K
d) Cs
Explanation: Cs, because of its low I.E. emits electron under the influence of even candle light.
6. Francium was discoverd by
a) Arfwedson
b) Davy
c) Perey
d) Bunsen & Kirchoff
Explanation: Francium was discoverd by Perey
7. Sodium is usually kept under
a) alcohol
b) kerosene oil
c) ammonia
d) water
Explanation:
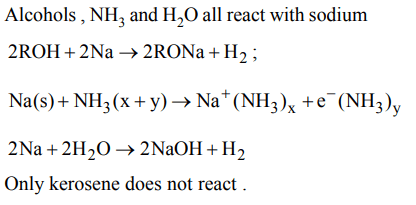
8. Sodium metal cannot be stored under
a) benzene
b) kerosene oil
c) alcohol
d) toluene
Explanation: Sodium metal cannot be stored under alcohol
9. Among \[KO_{2}, AlO_2^-, BaO_{2} and NO_2^+\] , unpaired electron is present in
a) \[ NO_2^+\] and \[BaO_{2}\]
b) \[KO_{2}\] and \[AlO_2^-\]
c) \[KO_{2}\] only
d) \[BaO_{2}\] only
Explanation:

10. The one with the largest size is
a) Na
b) \[Na^{+}\]
c) K
d) \[K^{+}\]
Explanation: The size increases down the group