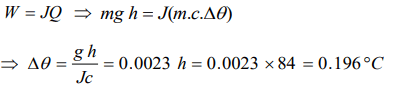1. If temperature scale is changed from °C to °F, the numerical value of specific heat will
a) Increases
b) Decreased
c) Remains unchanged
d) None of the above
Explanation:
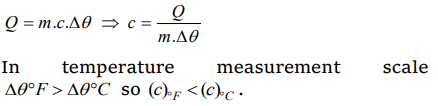
2. By exerting a certain amountof pressure on an ice block, you
a) Lower its melting point
b) Make it melt at 0°C only
c) Make it melt at a faster rate
d) Raise its melting point
Explanation: Increasing pressure lowers melting point of ice
3. When we rub our palms they gets heated but to a maximum temperature because
a) Heat is absorbed by our palm
b) Heat is lost in the environment
c) Produced of heat is stopped
d) None of the above
Explanation: Work done changes into heat energy, when the temperature of palm becomes above the atmosphere so it starts losing heat to the surroundings
4. A bullet moving with a uniform velocity v, stops suddenly after hitting the target and the whole mass melts be m, specific heat S, initial temperature 25°C, melting point 475°C and the
latent heat L. Then v is given by
a) \[mL=mS\left(475-25\right)+\frac{1}{2}.\frac{mv^{2}}{J}\]
b) \[mS\left(475-25\right)+mL=\frac{mv^{2}}{2J}\]
c) \[mS\left(475-25\right)+mL=\frac{mv^{2}}{J}\]
d) \[mS\left(475-25\right)-mL=\frac{mv^{2}}{2J}\]
Explanation:
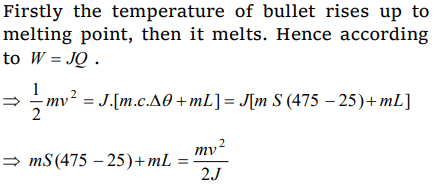
5. A water fall is 84 metres high. If half of the potential energy of the falling water gets converted to heat, the rise in temperature of
water will be
a) 0.098°C
b) 0.98°C
c) 9.8°C
d) 0.0098°C
Explanation:
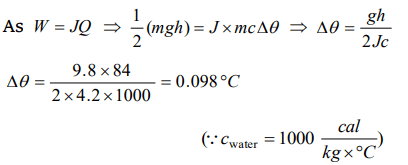
6. A body of mass 5 kg falls from a height of 30 metre. If its all mechanical energy is changed into heat, then heat produced will be
a) 350 cal
b) 150 cal
c) 60 cal
d) 6 cal
Explanation:
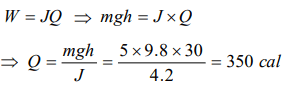
7. In supplying 400 calories of heat to a system, the work done will be
a) 400 joules
b) 1672 joules
c) 1672 watts
d) 1672 ergs
Explanation: W = JQ = 4.18 * 400 = 1672 joule
8.The weight of a person is 60 kg. If he gets 105 calories heat through food and the efficiency of his body is 28%, then upto how much height he
can climb (approximately)
a) 100 m
b) 200 m
c) 400 m
d) 1000 m
Explanation:
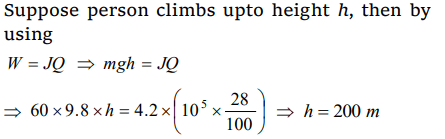
9. The temperature of Bhakhra dam water at the ground level with respect to the temperature at high level should be
a) Greater
b) Less
c) Equal
d) 0°C
Explanation: When water falls from a height, loss of potential energy causes rise in temperature.
10. The height of a waterfall is 84 metre. Assuming that the entire kinetic energy of falling water is
converted into heat, the rise in temperature of the water will be\[\left(g=9.8 m\diagup s^{2}, J =42 joule \diagup cal\right)\]
a) 0.196°C
b) 1.960°C
c) 0.96°C
d) 0.0196°C
Explanation: