1. The property utilized in the manufacture of lead
shots is
a) Specific weight of liquid lead
b) Specific gravity of liquid lead
c) Compressibility of liquid lead
d) Surface tension of liquid lead
Explanation: Surface tension of liquid lead
2.The dimensions of surface tension are
a) \[ \left[MLT^{-1}\right]\]
b) \[ \left[ML^{2}T^{-2}\right]\]
c) \[ \left[ML^{0}T^{-2}\right]\]
d) \[ \left[ML^{-1}T^{-2}\right]\]
Explanation:
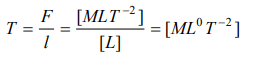
3. A wooden stick 2m long is floating on the surface of water. The surface tension of water 0.07 N/m. By putting soap solution on one side of the sticks
the surface tension is reduced to 0.06 N/m. The net force on the stick will be
a) 0.07 N
b) 0.06 N
c) 0.01 N
d) 0.02 N
Explanation:
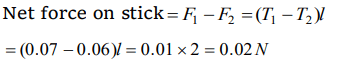
4. The force required to take away a flat circular plate of radius 2 cm from the surface of water, will be (the surface tension of water is 70
dyne/cm)
a) \[280\pi\] dyne
b) \[250\pi\] dyne
c) \[140\pi\] dyne
d) \[210\pi\] dyne
Explanation: Force required, F = 2 \[\pi\] rT = 2 \[\pi\] * 2 * 70 = 280 \[\pi\] Dyne
5.Surface tension may be defined as
a) The work done per unit area in increasing the surface area of a liquid under isothermal condition
b) The work done per unit area in increasing the surface area of a liquid under adiabatic condition
c) The work done per unit area in increasing the surface area of a liquid under both isothermal and adiabatic conditions
d) Free surface energy per unit volume
Explanation: The work done per unit area in increasing the surface area of a liquid under isothermal condition
6.Energy needed in breaking a drop of radius R into n drops of radii r is given by
a) \[4\pi T\left(nr^{2}-R^{2}\right)\]
b) \[\frac{4}{3}\pi \left(r^{3}n-R^{2}\right)\]
c) \[4\pi T\left(R^{2}-nr^{2}\right)\]
d) \[4\pi T\left(nr^{2}+R^{2}\right)\]
Explanation:
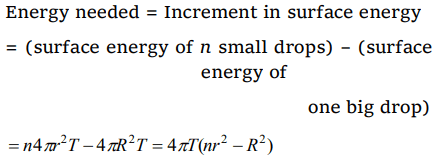
7.The potential energy of a molecule on the surface of liquid compared to one inside the liquid is
a) Zero
b) Smaller
c) The same
d) Greater
Explanation: Greater
8. Two droplets merge with each other and forms a large droplet. In this process
a) Energy is liberated
b) Energy is absorbed
c) Neither liberated nor absorbed
d) Some mass is converted into energy
Explanation:
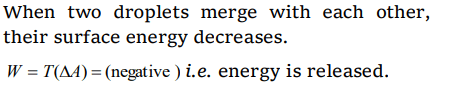
9. A drop of liquid of diameter 2.8 mm breaks up into 125 identical drops. The change in energy is nearly (S.T. of liquid =75 dynes/cm)
a) Zero
b) 19 erg
c) 46 erg
d) 74 erg
Explanation:
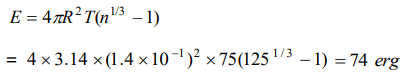
10. Radius of a soap bubble is 'r', surface tension of soap solution is T. Then without increasing the
temperature, how much energy will be needed to double its radius
a) \[4\pi r^{2} T\]
b) \[2\pi r^{2} T\]
c) \[12\pi r^{2} T\]
d) \[24\pi r^{2} T\]
Explanation:
